Understanding Your Fertilizer Types and Composition
Fertilization in forage systems can account for the large cost of maintaining pastures or increasing hay production. It is important to remember that all soils are not created equal and your forage productivity will depend on the soil’s capacity and chemical properties (especially pH) to supply nutrients. Forage nutrients requirements also depend on how much nutrients are being removed from the soil over time and how often those nutrients are replenished through a fertility management program.
Keep in mind that different forages have different nutrient contents. Nutrient removal, especially in hay production, can be estimated by using the average nutrient content of the forage species and the yield produced per acre.
Keep in mind that nutriment removal will increase as yield production increases. This means that over time the nutrient applications will be the greatest in forage and soils with the highest yields. That is also a reason why a soil test recommendation has a three-year plan.

Fertilizer use is an important factor for producing profitable forages and for maintaining soil nutrient balance over the years. The first step to developing a fertility program for your forage system must always start with a soil sample to determine nutrient needs. Most producers commonly make fertilizer applications without any soil nutrient information, which means the possibility of over-fertilizing or under-fertilizing. Either approach will have an economic impact on your farm. Nitrogen, phosphorus, and potassium are called primary macronutrients because they are needed in large quantities to support forage production at profitable economic levels and to replace nutrients removed with each hay harvest.
Fertilizer Functions
Most fertilizer suppliers will have different fertilizer types available depending on the source, region, and environmental conditions for utilizing those fertilizer types. By understanding the basics behind fertilizer labels, you can choose the right fertilizer type(s) for your forage system based on soil nutrient availability, forage crop, and expected yields.
Nitrogen and potassium are usually required in larger quantities on forage systems than phosphorus because they can be taken up in larger quantities by the forage crop or lost through different processes. Phosphorus tends to be very stable in the soil and it is required in lower quantities than N and K.
Nitrogen is needed for biomass production, increase leaf growth, and increase crude protein. Phosphorus (P) is essential for seed germination, strong root growth, and seed development. Potassium (K) enhances overall growth while helping to regulate root and top growth and in-creasing plant protection to pest resistance and drought and cold tolerance.
Calculating Your Fertilizer Nutrient Content
All fertilizer types must provide uniform information that will help forage producers to compare products. Every bag of fertilizer contains three names or abbreviations in the following order Nitrogen (N), Phosphorus (P2O5), and potassium (K2O) (Fig. 1). They represent the oxide form for elemental phosphorus (P), and potassium (K). These can be converted to elemental P (P = P2O5 x 0.44 or P2O5 = P X 2.3) or elemental K (K = K2O x 0.83 or K2O = K x 1.2). These letters are also associated with three numbers that represent an N-P-K ratio and the proportion of each nutrient in the same order (N-P-K). Fertilizer ratio is the ratio of weight percent of N-P2O5-K2O and is determined by dividing the three numbers by the smallest of the three. Fertilizer manufacturers usually produce different fertilizer grades depending on the market (turf vs. forages). For example, a bag of fertilizer with a 34-46-60 means that a 100-bag contains 34% nitrogen, 46% phosphorus, and 60% potassium by weight. In order words, that 100-bag contains 34 lbs of nitrogen (0.34 x 100 = 34), 46 lbs of phosphorus (0.46 x 100 = 46), and 60 lbs of potassium (0.60 x 100 = 60). Usually, these fertilizer grades can be formed by using only source (34-0-0, 46-0-0, 0-46-0, 0-0 -60) or by mixing two or more nutrient sources to form a blend (0-20-20, 18-46-0, 33-0-0S). Fertilizer blends are usually called "mixed fertilizers” and they contain particles of more than one color.
Selecting a Fertilizer Grade for Forage Production
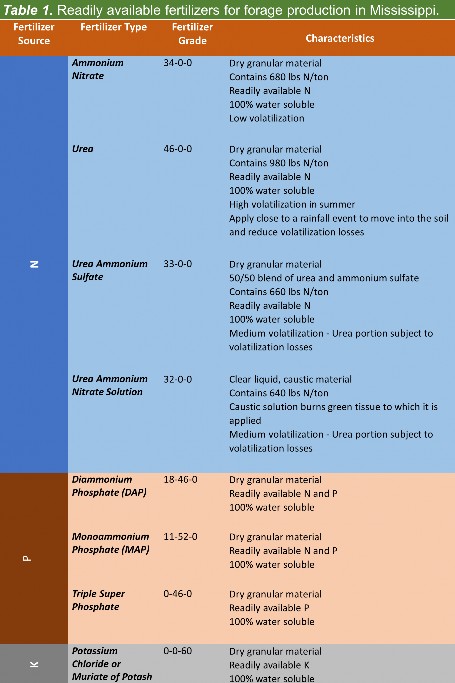
The best way to select a fertilizer grade is to have your soil tested. Soil test all fields on a 2 (hay) to 3-year (pasture) interval. Soil samples should be collected at the same time during the year to avoid seasonal variation in soil pH levels and nutrient availability. Phosphorus and potassium fertilizers should be applied according to soil test recommendations (Table 1). These recommendations provide for building soil fertility levels when soil samples show that the nutrient levels are low and maintaining high levels when the high fertility has been achieved. In general, nitrogen fertilizer application to pastures or hay should be split to increase efficiency. It is recommended that to apply 60 units of N per acre in split applications (after green-up) and mid-summer after the second grazing cycle. For hay production, nitrogen applications of 50 units per acre per cut of hay should be sufficient when hay is cut in a 28 to 35-day cycle. However, N applications in the summer should be carefully timed with plenty of moisture along with choosing the correct nitrogen source to reduce losses. Producers cannot overcome a lack of rainfall with increased fertilization. Table 1 provides some information about the types of nitrogen sources that are available for use in forage production in Mississippi.
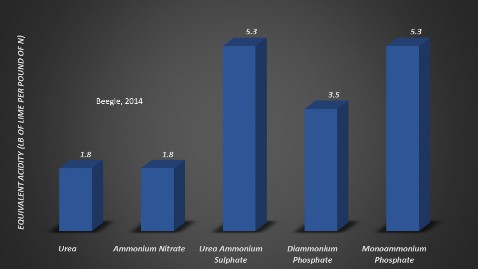
Sometimes producers are concerned with the production of acidity by the reaction of fertilizer materials, especially from N fertilizers. The acidity of a fertilizer is usually expressed as the amount of pure lime that would be required to neutralize the acidity produced by the reaction of the fertilizer. Figure 2 indicates that the highest amount of acidity is not necessarily created by nitrogen source, but amount and number of applications. Although phosphate-based fertilizer has high- er acidity, they are applied less often and in smaller quantities than N based fertilizers.
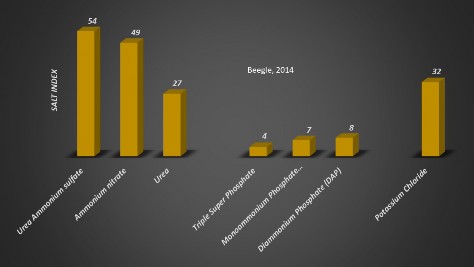
Another property to consider when selecting a fertilizer source is the amount of soluble salt in the fertilizer. A high concentration of soluble salts can cause plant injury and prevent seed germination. Application of fertilizers at the recommended rates usually do not have high levels of soluble salts to cause plant injury; however, when fertilizer is mixed with seed for planting, soluble salt levels that are high enough can cause seed embryo damage and reduce germination. The salt index of several fertilizer types is shown in Figure 3.
Finally, the selection of a nitrogen fertilizer source should be based on the cost per pound of N and the potential for volatilization losses. For example, using local fertilizer prices for urea ($382/ton, $0.61/lb N), ammonium nitrate ($350/ton, $0.51/lb N), urea ammonium sulfate ($415/ton, $0.63/lb N), and 32% UAN solution ($294/ton). This means that the cost per acre for applying 50 lbs N/ac will be $30.50 for urea, $25.50 for ammonium nitrate, and $31.50 for urea ammonium sulfate, and $23.0 for UAN solution. In many situations, the cheapest fertilizer cost or the fertilizer with a higher N grade might not be the best option. It is also important to consider weather conditions (moisture, heat, and humidity) that can impact the potential for volatilization losses. For example, urea can have volatilization losses as high as 20 to 30% in warm temperatures with no rainfall at application time or soon after. A reason why urea is more suited for applications to cool-season grasses during the winter, spring, and late fall.
Summary
Developing a well-managed and efficient forage fertilization program that can increase profitability routinely depends on soil testing as well as maintain records of the applied fertilizers and nutrient removal. The amount of nitrogen to be applied per unit should be based on expected yields and the rainfall pattern during the season along with split applications to increase utilization and efficiency. It is also important to select the appropriate fertilizer source(s) to supply the needed nutrient at a lower cost.


